Related Technical Articles
Metals (ferrous & non-ferrous) are susceptible to environmental physical, chemical, or electrochemical deterioration when these come in contact with moisture and oxygen. Metals form an oxide layer on the surface which indirectly protects metal if oxide is denser than the parent metal. Only oxygen or only water cannot cause corrosion of the metals, both constituents must be present together to cause corrosion.
There are plenty of technologies available to protect metals from atmospheric corrosion such as barrier coatings like paint, plastic, and powder, peelable coatings, rust preventive oils, cathodic protection, dip galvanization, etc. restricting metal surfaces from acidic vapors, chloride, bromide corrosion. The major problems arise while the packaged part or equipment is unpacked. The paint, oil, or coating leaves a residue while cleaning [1].
Polyethylene film offers many advantages as a packing material. Polyethylene packaging in conjunction with Vapor Corrosion Inhibitors (VCI) is the most effective solution to metal jobs in the transportation industry. The impermeable morphology of the film makes packaged objects free from water vapor, oxygen in combination with most other contaminants in the atmosphere. However, the diffusion of VCI molecules into the headspace of the package became a concern when it encounter a high level of acid gas contaminations. The film is available in markets with different sizes ranging from 25-150 µm thickness and combination with metalized aluminum layers (multilayer films). Conventional polyethylene films are more permeable to water vapor, acid, and other gas constituents than the multilayer films. The diffusivity varies with thickness and the type of polymeric materials. In addition to ease of handling, it is also fully recyclable.
VCIs are organic compounds that include primary, secondary amines, and azole compounds. Cyclohexyl ammonium carbonate (CHAC), diisoporopylammonium nitrite (DIPAN) and dicyclohexyl ammonium nitrite (DICHAN)[2], Benzotriazoles [3], 6-methoxy-aminobenzothiazole derivatives[4], 1,6-diaminohexane [5] for Al, Zn, Mild Steel (MS). The parent compounds have specific vapor pressure under ambient conditions to essentially cover the surface of the metal by adsorption. In presence of moisture, the VCI active becomes polarized, and is attracted towards anode or cathode of the metal which protects the metal corrosion from the electrochemical process by adsorbing ions on the surface. Among these chemistries, primary amines are ecologically safe, non-toxic in nature, and are also admissible in REACH compliance.
Materials & Methods
Testing Methods
Razor Blade Test:
German VIA TL 8135-002 Method 010 (Short term):
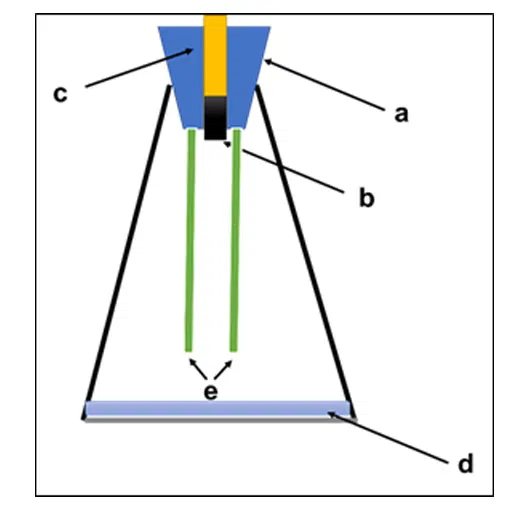
Fig 1: K-Test experimental setup a) Rubber bung, b) Test Metal sample, c) Glass tube with frozen water d) Glycerin water mixture e) VCI-OS film
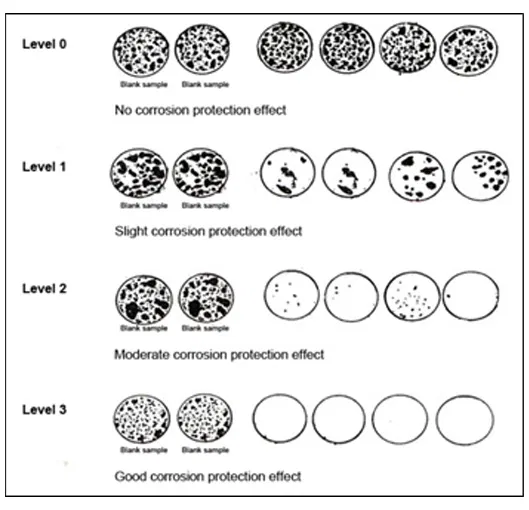
Fig 2: German VIA Test Rating
German VIA TL 8135-002 Method 010 (Short term):

Fig 3: German APL 004 VIA Test setup
ASTM B 117 (Salt Fog test):
Results and Discussions
Razorblade Test:


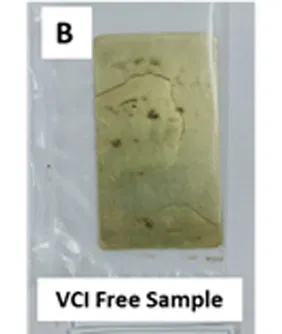

Fig 4: Razor Blade test on A) MS & B) Brass
German VIA APL 010:


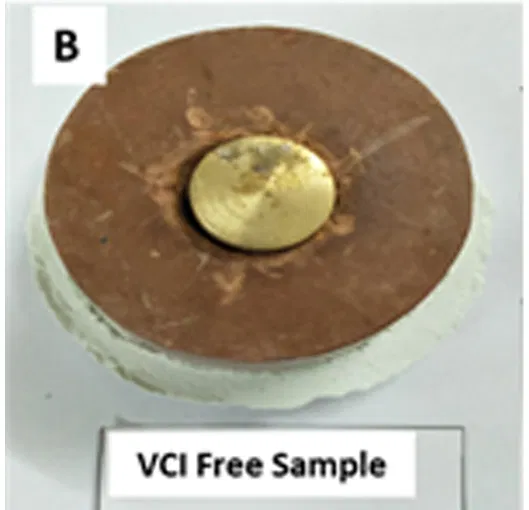

Fig 5: German VIA APL 010 test study on A) MS, B) Brass
German VIA APL 004:




Fig 6: German VIA APL 004 test study on A) MS, B) Brass Salt fog Test (ASTM B-117):




Fig 7: Salt Fog test 500 hours exposure observations A) MS, B) Brass
Conclusion
About NICHEM
Long-standing Specialty Chemicals player with ISO 9001:2015 certification and a history of providing specialty solutions for over 25 years. The company is headed by senior chemical industry specialists with the combined expertise of more than 100 years. With an emphasis on eco-friendly, non-toxic products, the company’s primary strength is research, development, and customization. More information on NICHEM can be found at https://nichem.solutions.
References:
[1] Donald A. Kubik, New Technology for Vapor Corrosion Inhibiting (VCI) Transparent Plastic Packaging Combined with Acid-gas Scavenging (AGS), NACE international-corrosion conference and expo, 2010
[2] U. Kunzelmann, H.J. Jacobasch, G. Reinhard, Werkst. Korros. 40, 723, 1989.
[3] B.A. Miksic, M. Tarvin, G.R. Sparrow, NACE Corrosion 89, New Orleans, USA, April 4–8, paper 607, 1989, paper 607
[4] W. Skinner, Corros. Sci. 35,1491, 1993.
[5] M.A. Quraishi, D. Jamal, Corrosion 58, 387, 2002.
[6] Alla. Y. Furman, Performance and Testing of Vapor Phase Corrosion Inhibitors, Corrosion, paper 04418, 2004
[7] Nelson cheng, Ascertaining the Effectiveness of VCI Anti-Corrosion Properties of Vappro VCI Inhibicard Coated with Vappro MBL2200 Using German Test Method TL 8135-002, research gate,2016.
[8] Michel prenosil, Volatile corrosion inhibitor coatings, Supplement to Materials Performance, 2001.



