Related Technical Articles
Silicon (Si) is a chemical element having symbol Si atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster (1).
Silica (SiO2) is a compound of Silicon Oxygen is represented as SiO2 (Silicon Dioxide). It is amorphous in nature. Its crystalline varient is quartz (2).OrthoSilicic Acid (OSA) is the simplest soluble form of Silica (SiO2). It is an inorganic compound of Silica (SiO2) Silicates (SiO44-) a precursor to other Silicates. It has Silicon (Si) tetrahedrally coordinated to 4 hydroxyl (OH) groups. It is represented by the chemical formula Si (OH)4 or H4O4 Si. It is a weak acid. Its IUPAC name is Silicic Acid (4).In the earth’s crust, more than 90% Silicon (Si) occurs in the forms of Silica (SiO2) or Silicates (Salts in which the anion contains both Silicon Oxygen – SiO (SiO44-). It constitutes to 28% of the earth’s crust by mass.
It is also present in ocean water as Orthosilicic acid (Si(OH)4) or its conjugate bases (Orthosilicate Ions). In ocean water, the algae known as diatoms regulate the biogeochemical
cycles of silica. These algae polymerize the Silicic Acid to Biogenic Silica (Amorphous Silica) to form their cell walls (3, 4).
Silicic Acid (OSA) has been reported to play important roles in biomineralization. OSA is of particular interest in agriculture as it is reported to be the most ‘assimilable’ or the
‘bioavailable’ form in which the plants uptake Silicon (Si) from the soil. The importance of Silicon (Si) in agriculture is very well documented for the last few years. It helps plants to
manage biotic and abiotic stress and enhance output. It is proposed to be working at a cellular level to impart benefits to plant cells at very low ppm (parts per million) concentration. We have found that every part of a living plant, the roots, trunk, branches , leaves, flowers, seed, fruit contain detectable concentration of silica.
Silicic Acid does not exist in nature. It has to be chemically synthesised using Silicates and Acid. It was first synthesised by Sir Thomas Graham (1861) by dialysing dilute silica sols
obtained by mixing an aqueous solutions of sodium silicate with hydrochloric acid. Nowadays, it is manufactured using Patrick’s process which consists essentially of gelation of an alkali metal silicate by means of acid. Monomeric and Polymeric Silicic Acids are formed by neutralising a solution of sodium metasilicate or water glass by acid. The condensation reaction of monomeric or polymeric silicic acid can be according to the following scheme (6). Fig 1 depicts the polymerisation of silicic acid.
(HO)3SiOH + HOSi(OH)3 (HO)3Si-O-Si(OH)3 + H2O
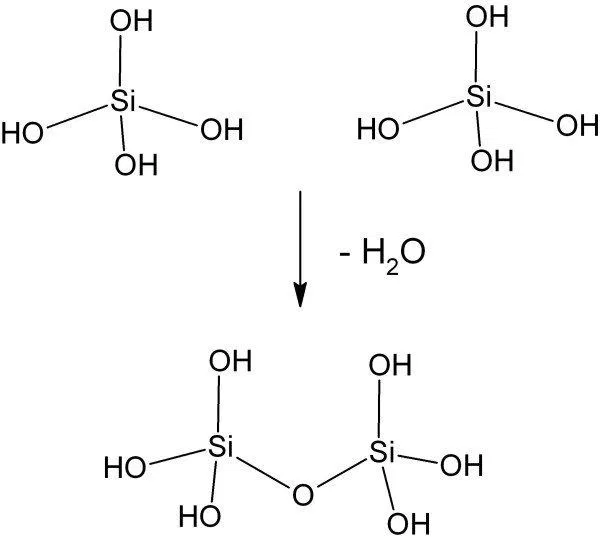
Fig 1: Polymerisation of silicic acid molecules through formation of siloxane bond and water(5)
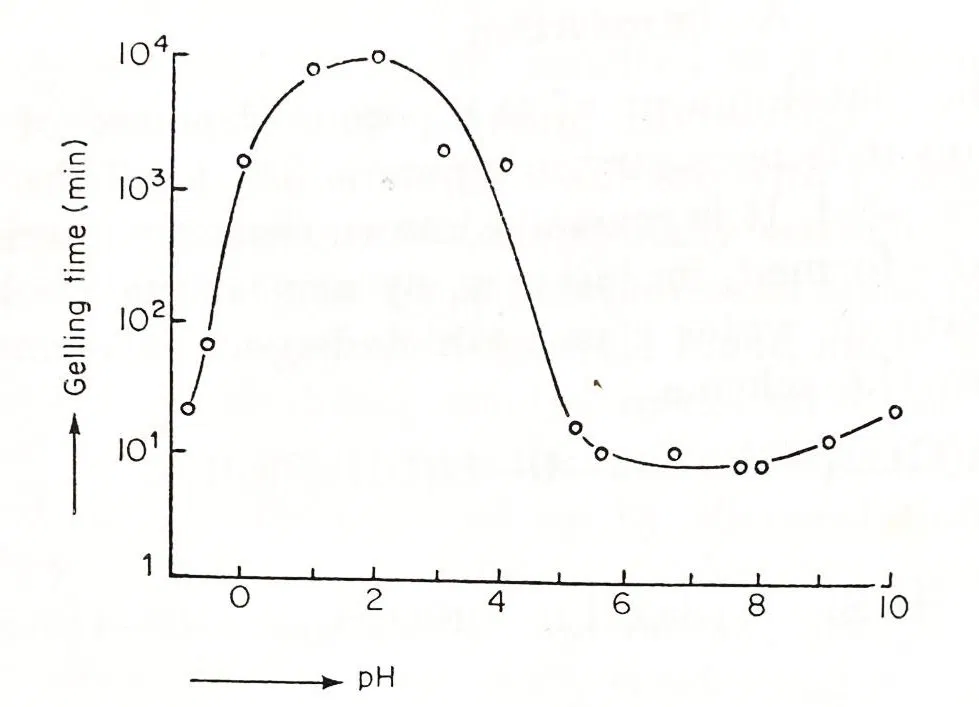
Fig 1: Gelling time of a silica sol as a function of pH. SiO2 concentration:5% (pH 1-5, pH 8-10), 2.5% (pH 5-8) (6)
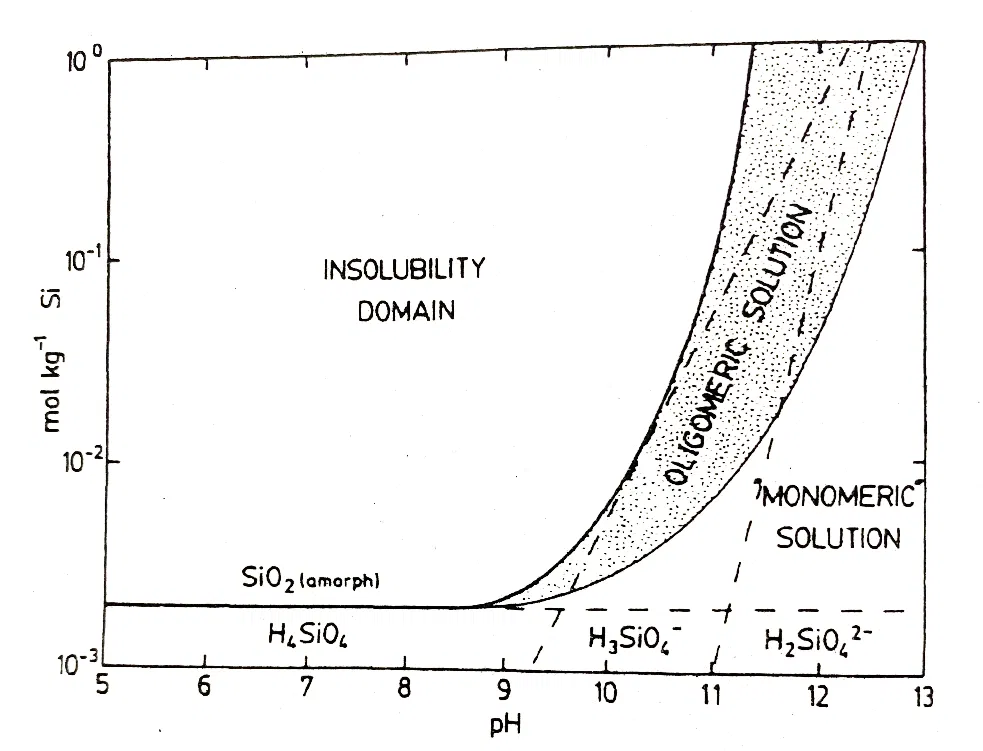
Fig 2: Stability Diagram for Soluble Silicates (7)

Fig 3: Mechanisms of silica precipitation (8)
This process of stabilization of OSA in CropSil is patented in India, USA & Canada.
To know more, please visit our website www.nichem.solutions
About NICHEM
Long-standing Specialty Chemicals player with ISO 9001:2015 certification and a history of providing specialty solutions for over 25 years. The company is headed by senior chemical industry specialists with the combined expertise of more than 100 years. With an emphasis on eco-friendly, non-toxic products, the company’s primary strength is research, development, and customization. More information on NICHEM can be found at https://nichem.solutions.
References:
1. Silicon (https://en.wikipedia.org/wiki/Silicon#Applications)
2. Silicon Dioxide (https://en.wikipedia.org/wiki/Silicon_dioxide#Natural_occurrence )
3. Silicate (https://en.wikipedia.org/wiki/Silicate)
4. Ortho-silicic Acid (https://en.wikipedia.org/wiki/Orthosilicic_acid)
5. Dorota Napierska et al, The Nanosilica Hazard:Another Variable Entity; Particle &
Fibre Toxicology, Dec 2010
6. BJ Linsen et al, Physical Chemical Aspects of Adsorbants and Catalysts, Chapter 5 –
Porous Silica, 1970, Pg 216
7. LE Datnoff et al, Silicon in Agriculture: Chapter 4 – A primer on the aqueous chemistry
of silicon, 2001, Pg 63-64
8. Sebastian Wilhelm and Matthias Kind, Influence of pH, Temperature Sample Size on
Natural Enforced Syneresis of Precipitated Silica; Polymers 2015,7, Pg 2505-2506


