Related Technical Articles
Durable antimicrobial protection has become an essential requirement across many industries. Polymer-enabled antimicrobial technologies offer improved durability and reduced leaching compared to conventional additives. The effectiveness of these systems depends largely on polymer chemistry, which determines how antimicrobial functionality is delivered and maintained.
Antimicrobial Polymers: An Overview
Antimicrobial polymers are materials where the biocidal functionality is chemically bonded into the polymer chain itself. To design high-performance systems for medical, food, and industrial applications, formulators must move beyond “one-size-fits-all” additives. Unlike traditional antimicrobial additives that are physically blended and may leach out over time, these polymers provide a permanent functional surface.
Key Application Sectors
Hygiene oriented products have led to an intense demand for non-leaching and durable antimicrobial additives.
Antimicrobial polymers are utilised across diverse industries to prevent biofilm formation and cross-contamination.
- Water Treatment and Membranes: Used in filtration systems to inhibit the growth of bacteria and algae on membrane surfaces. This reduces fouling and extends the operational life of the filtration units.
- Personal Care and Cosmetics: Integrated into packaging and formulations to prevent microbial spoilage. This ensures product safety and shelf stability without excessive chemical
- Food-Contact Materials and Packaging: Applied in films and containers to slow down the growth of foodborne pathogens. This helps in maintaining food quality and reducing waste throughout the supply chain.
- Medical and Hygiene Surfaces: Essential for hospital equipment, surgical tools and high-touch These materials provide a continuous kill rate against Healthcare-Associated Infections (HAIs).
- High touch surfaces: High touch surfaces refers to objects like handles/railings/seats of public transport that are frequently handled or contacted by multiple individuals increasing the likelihood of transfer of contaminants and microorganisms.
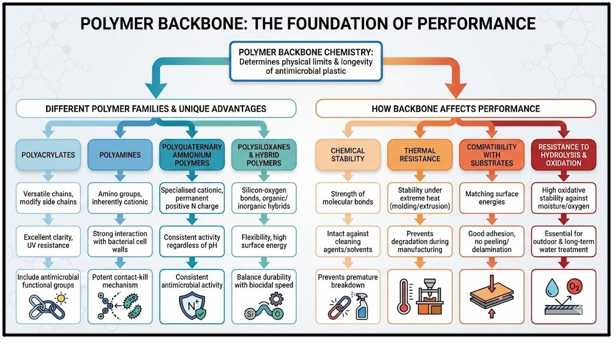
Fig 1. Polymer Backbone: The Foundation of Performance
Charge Density and Ionic Functionality
The effectiveness of many antimicrobial polymers relies on the interaction between the polymer’s surface and the microbial cell membrane.
Cationic Groups in Antimicrobial Action
Specific functional groups are engineered into the polymer structure to provide the necessary ionic attraction to disrupt pathogens.
- Quaternary Ammonium Groups: These are the most common cationic agents. They feature a central nitrogen atom with four organic groups This creates a permanent positive charge that pierces bacterial cell walls.
- Phosphonium Groups: These utilise a phosphorus atom as the cationic centre. They often exhibit higher thermal stability than ammonium counterparts. This makes them suitable for high-temperature polymer processing.
- Guanidinium Groups: These groups contain multiple nitrogen atoms that distribute the positive charge. They are highly effective at penetrating complex biofilm structures and resistant bacterial strains.
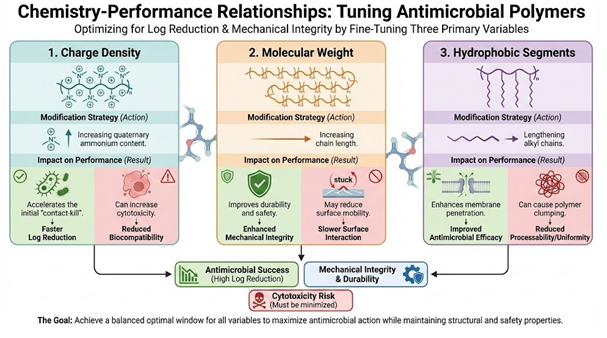
Fig 2. The Impact of Chemical Modifications on Antimicrobial Polymer Design and Performance
Performance Relationships
The physical behaviour of the polymer is directly linked to the concentration and type of ionic charge present in the matrix.
- Charge Density: This refers to the number of positive charges per unit area or molecular weight. Higher charge density generally leads to faster kill rates. However, if the density is too high, it can cause the polymer chains to repel each other. For applications like wearable devices or medical textiles, molecular weight and charge density is optimised to prevent the polymer from penetrating deep into the human skin layer.
- Microbial Membrane Disruption: When a negatively charged microbe contacts a cationic surface, the ionic attraction draws the microbe toward the polymer. The long alkyl chains attached to the cationic centres then insert themselves into the lipid This causes the cell to leak and eventually rupture.
- Selectivity vs. Cytotoxicity: Cationic polymers must be designed to target the specific lipid composition of bacterial membranes. If the charge is too aggressive or non-selective, the material may exhibit cytotoxicity. This means it could harm healthy human tissue upon
Molecular Weight and Chain Architecture
The physical dimensions and geometric arrangement of polymer chains determine how effectively the material interacts with microorganisms.
Low vs. High Molecular Weight Polymers
The length of the polymer chain directly influences its physical behaviour and biocidal speed.
- Mobility: Low molecular weight polymers possess higher chain This allows them to diffuse more rapidly through a matrix to reach the surface. Conversely, high molecular weight polymers are bulky and move slowly, which can limit their initial antimicrobial response.
- Surface Interaction: High molecular weight polymers often provide better surface Because the chains are longer, they can form more contact points with a microbe’s cell wall. This increases the likelihood of membrane disruption upon contact.
- Diffusion Limitations: In thick plastic parts, high molecular weight actives can become trapped deep within the bulk. This creates a diffusion limitation where the interior contains the active agent, but the surface remains unprotected.
Linear, Branched, and Crosslinked Architectures
The shape of the polymer molecule dictates how it packs together and how it presents its active sites.
- Linear Structure: These consist of a single long chain. They provide high flexibility and are easier to process. However, they may be prone to leaching if they are not chemically anchored to the substrate.
- Branched Structure: These contain side chains that extend from the main backbone. Branching increases the surface area of the molecule. This allows for a higher concentration of active functional groups to be exposed to the environment.
- Crosslinked Structure: These form a three-dimensional network where chains are chemically bonded to one This architecture is the most durable. It creates a permanent, non-leaching surface that is resistant to mechanical wear and chemical cleaning.
Impact on Performance and Durability
The combination of weight and structure determines the lifecycle and safety profile of the antimicrobial product.
- Contact-Kill Efficiency: Crosslinked and high molecular weight polymers rely on a “contact-kill” mechanism. The chain architecture must be engineered to ensure that enough active sites are available on the outermost layer to maintain high log reductions.
- Migration and Leaching Behaviour: Low molecular weight linear polymers are more likely to migrate. While this can provide a “zone of inhibition” around the product, it also means the antimicrobial effect will eventually be exhausted.
- Durability of Antimicrobial Effect: Crosslinked networks provide the highest durability. As the active sites are locked into a stable matrix, they do not wash away or volatilize. This ensures that the plastic remains functional for years, even in high-traffic or harsh industrial
Hydrophilic - Hydrophobic Balance
The efficacy of an antimicrobial polymer is governed by its amphiphilicity, which is the balance between water-attracting (hydrophilic) and water-repelling (hydrophobic) regions.
Microbial cell membranes are composed of lipid bilayers. Hydrophobic domains on the polymer chain act as molecular needles. They partition into these fatty layers, causing structural failure and cytoplasmic leakage.
Importance of Hydrophilicity
A polymer that is purely hydrophobic would be impossible to use in most industrial applications. Hydrophilic groups are necessary for functional integration.
- Water Solubility: In water treatment and personal care, the polymer must be soluble or dispersible in aqueous phases to interact with suspended pathogens.
- Processability: Hydrophilic groups improve the ease of handling during the manufacturing of coatings and films.
- Uniform Dispersion: Within a plastic matrix, hydrophilicity helps the antimicrobial agent distribute evenly. This prevents the formation of “dead zones” where no antimicrobial protection exists.
Tuning Amphiphilicity for Optimal Performance
The “Goldilocks zone” for antimicrobial activity lies in the precise ratio of these two domains.
- Optimal Antimicrobial Performance: If a polymer is too hydrophilic, it will stay in the surrounding water and fail to bind to the microbe. If it is too hydrophobic, the polymer chains will clump together (aggregate) to hide from the water, making the active sites
- Tuning the Balance: Formulators adjust the length of alkyl chains (hydrophobic) relative to the number of ionic groups (hydrophilic). This tuning ensures the polymer is mobile enough to reach the target but aggressive enough to disrupt the membrane upon arrival.
Functional Group Placement and Accessibility
The concentration of biocidal groups in a polymer matrix does not guarantee antimicrobial efficacy. For a surface to be truly contact-active, the antimicrobial moieties must be physically accessible to the target microorganism.
Surface Availability of Antimicrobial Moieties
Surface availability refers to the percentage of active sites that reside on the outermost layer of the plastic. In non-migratory systems, any active group buried within the bulk of the material remains inert. If the polymer chains do not orient themselves to present the cationic centres outward, the material will fail standardised tests despite having high loading levels.
Pendant Groups vs. Backbone-Incorporated Actives
The choice of where to place the antimicrobial group on the molecular chain impacts both the biocidal speed and the mechanical strength of the polymer.
Feature | Pendant Groups (Side-Chain) | Backbone-Incorporated |
Structure | Active sites are attached as side chains. | Active sites are part of the main chain. |
Accessibility | High, groups extend outward like “brushes.” | Moderate; often shielded by the chain. |
Charge Density | Can be very high without affecting the backbone. | Limited by polymer structural integrity. |
Durability | Higher risk of side-chain cleavage. | Extremely robust and permanent. |
Processing | Easier to modify existing polymers. | Requires de novo monomer synthesis. |
Steric Effects and Polymer Conformation
Steric hindrance occurs when bulky molecular structures physically block the active site from contacting the microbe.
- Molecular Shielding: If the alkyl chains or side groups are too large, they can fold over the cationic centres. This “masking” effect prevents the antimicrobial moiety from reaching the bacterial cell wall.
- Conformational Changes: Polymers change their shape based on the surrounding environment. In dry conditions, a polymer may collapse into a dense coil, hiding its active sites. Upon exposure to moisture (aqueous bacterial suspensions), the chains must expand to expose the biocidal groups for effective kill rates.
Mechanism of Action: Contact-Kill vs. Controlled Release
The functionality of antimicrobial polymers is categorised by how the active agent interacts with the environment. Engineers must choose between a system that kills upon physical contact or one that releases biocides into the surrounding area.
Comparative Mechanisms of Antimicrobial Action
The table below summarises the technical differences between non-leaching and controlled-release architectures.
Feature | Contact-Kill (Non-Leaching) | Controlled-Release (Leaching) |
Primary Mechanism | Physical disruption of the cell envelope. | Chemical poisoning of microbial pathways. |
Agent Movement | Remains chemically tethered to the polymer. | Diffuses out of the polymer matrix. |
Duration | Permanent; lasts for the life of the plastic. | Temporary; ends when the reservoir is empty. |
Zone of Inhibition | None; kills only what touches the surface. | Large; kills microbes in the vicinity. |
Best Use Case | Medical implants and high-touch surfaces. | Water treatment and food packaging. |
Non-Leaching Contact-Active Polymers
These systems utilise a “mechanical” kill strategy. The polymer does not lose mass over time. Instead, it acts as a permanent biocidal shield.
- Membrane Disruption: Cationic groups like quaternary ammonium or phosphonium act as molecular “spears.” They create an ionic attraction that pulls the negative microbial membrane toward the surface. The hydrophobic segments then pierce the lipid This causes immediate loss of cytoplasmic pressure and cell death.
- Protein Denaturation: Certain polymer backbones can interact with the surface proteins of a virus or bacteria. This interaction unfolds the protein structure. It renders the microbe unable to replicate or infect a host.
Controlled-Release Polymer Systems
These systems act as reservoirs. They are designed to maintain a “kill zone” around the plastic part to prevent biofilm formation before it starts.
- Diffusion-Controlled: The antimicrobial agent is physically trapped within the polymer matrix. Over time, moisture or a concentration gradient pulls the agent to the surface and into the environment. Fick’s Laws of Diffusion govern the rate of release.
- Degradation-Triggered: The polymer backbone itself is biodegradable or erodible. As the plastic breaks down due to hydrolysis or enzymatic activity, it releases the encapsulated biocides. This is commonly used in agricultural films and speciality medical sutures.
Compatibility with Formulations and Substrates
The final efficacy of an antimicrobial polymer depends on its integration with co-additives and the target substrate. Chemical interference can significantly alter the “effective” charge density of the material.
Interactions with Co-additives: Anionic surfactants or salts often neutralise cationic active sites. This leads to precipitation or a total loss of biocidal activity. Conversely, non-ionic additives generally improve dispersion without interfering with the ionic charge.
Substrate Performance:
- Plastics and Fibres: Requires high thermal stability and uniform melt-blending to ensure surface bioavailability.
- Coatings and Aqueous Systems: Need precise chemical adhesion or solubility to maintain a stable functional layer.
Environmental Influence on Performance
Factor | Impact on Antimicrobial Activity |
pH Levels | Controls the ionisation state; acidic pH typically enhances cationic activity. |
Ionic Strength | High salt concentrations screen charges, reducing electrostatic attraction to microbes. |
Temperature | Higher heat increases chain mobility but may trigger polymer degradation. |
Durability, Wash-Off Resistance, and Long-Term Efficacy
The commercial viability of an antimicrobial polymer is measured by its ability to remain active under rigorous service conditions.
Polymer Anchoring
This involves chemical grafting or crosslinking the antimicrobial chain to the host substrate. This covalent bonding prevents the active sites from being stripped away during mechanical or chemical stress.
Resistance and Lifecycle Performance
| Stress Factor | Impact on Efficacy |
| Repeated Washing | Non-leaching polymers resist detergent stripping, maintaining log reduction through hundreds of cycles. |
| Abrasion | Crosslinked architectures ensure that, as the top layer wears down, new active sites are exposed. |
| Environmental Stress | High-performance backbones resist UV degradation and oxidation, preventing “active site masking.” |
Lifecycle Correlation
The durability of the antimicrobial effect is a direct result of the backbone’s chemical stability and the strength of its attachment to the substrate. Properly engineered polymers ensure that biocidal performance remains consistent from the first use until the end of the product’s life.
Safety, Toxicology, and Regulatory Considerations
The industrial adoption of antimicrobial polymers is strictly governed by global safety standards to prevent human exposure and environmental toxicity. Regulatory bodies differentiate between “treated articles” and “biocidal products” based on the material’s chemical behaviour and migration potential.
Global Regulatory Perspectives
The legal classification of an antimicrobial polymer depends on its intended use and its mobility within the matrix.
| Agency | Regulatory Focus |
| EPA (USA) | Regulates polymeric actives under FIFRA; distinguishes between product protection and public health claims. |
| FDA (USA) | Governs food-contact and medical materials; focus is on Migration Limits (SML) and biocompatibility. |
| ECHA (EU) | Enforces the Biocidal Products Regulation (BPR); requires data on environmental persistence and bioaccumulation. |


